Comparative Molecular Modelling Studies of Coumarin Derivatives as Potential Antioxidant Agents
DOI:
https://doi.org/10.5530/fra.2017.1.5Keywords:
Antioxidant activity, Coumarin, DPPH, HOMO, LUMO, H2O2Abstract
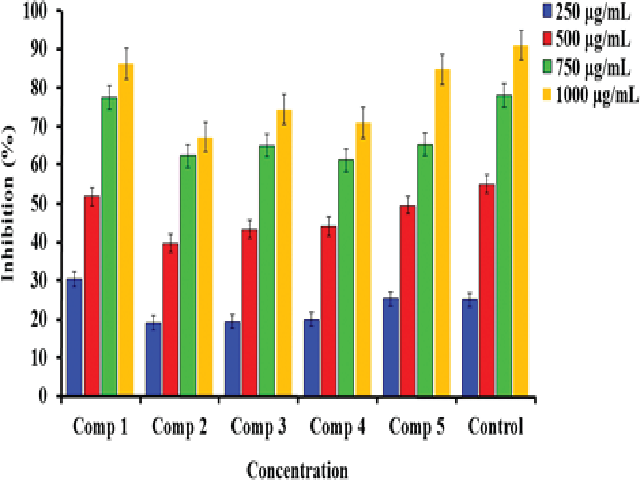
Background: Coumarin is quite extend in plants including various vegetables, flavors, foods. Coumarin has various significant medicinal activities such as anti-inflammatory, anti-oxidant, anti-viral, anti-microbial, anti-cancer and has been indicated to increase central nervous system activity. Methods: The syntheses of tailor-made coumarins, which are highly compelling for medical applications due to their structurally interesting antioxidant activity, were report in this manuscript. Coumarins were synthesized successfully through the modification of 4-hydroxycoumarin by different reaction steps. The molecular structures of the coumarins were characterized by the Fourier transformation infrared and Nuclear magnetic resonance (NMR) spectroscopies. The antioxidant efficiency of the 4-hydroxycoumarins were evaluated by typical spectroscopic method, using radicals DPPH• and H2O2. Results: The new coumarins synthesized in this work exhibited an excellent antioxidant compared to the free vitamin C. Molecular modelling studies using DFT (Density Functional Theory) calculations showed that there is a high correlation between dipole moment, Ionization potential (IP), Electron affinity (EA), Hardness (η), Softness (S), Electro negativity (μ), energy gap, HOMO (Highest Occupied Molecular Orbital) and LUMO (Lowest Unoccupied Molecular) energies and antioxidant activity. Conclusion: New coumarin derivatives were successfully synthesized using chemical methods. The characterized of these coumarins were done by using different spectroscopic techniques (FT-IR and NMR) and micro-elemental analysis (CHNS). The antioxidant activity of these coumarins were determined by using DPPH and hydrogen peroxide assays. Results indicated that the new coumarins possess higher scavenging activity than vitamin C.
Downloads
Metrics